CAR T-Cell Therapy: Implications for Navigation Practice
A Resource Guide for the Navigator
Academy of Oncology Nurse & Patient Navigators: Mission and Vision
The mission of the Academy of Oncology Nurse & Patient Navigators (AONN+) is to advance the role of patient navigation in cancer care and survivorship care planning by providing a network for collaboration and development of best practices for the improvement of patient access to care, evidence-based cancer treatment, and quality of life during and after cancer treatment. Cancer survivorship begins at the time of cancer diagnosis. One-on-one patient navigation should occur simultaneously with diagnosis and be proactive in minimizing the impact treatment can have on quality of life. In addition, navigation should encompass community outreach to raise awareness targeted toward prevention and early diagnosis, and must encompass short-term survivorship care, including transitioning survivors efficiently and effectively under the care of their community providers.
The vision of AONN+ is to increase the role of and access to skilled and experienced oncology nurse and patient navigators so that all patients with cancer may benefit from their guidance, insight, and personal advocacy.

AONN+ would like to acknowledge the efforts and dedication of oncology nurse navigator Peg Rummel, RN, MHA, OCN, NE-BC, HON-ONN-CG, who dedicated her knowledge, time, and efforts to enhance the care of patients with cancer through the development of this guide.

The endorsement mark certifies that the information presented in educational seminars, publications, or other resources is reliable and credible.

How to Use This Resource
This resource is designed for oncology nurse and patient navigators who are caring for patients with multiple myeloma undergoing chimeric antigen receptor (CAR) T-cell therapy. This guide provides information that might be useful to other personnel as well, who may be associated with the CAR T-cell program.
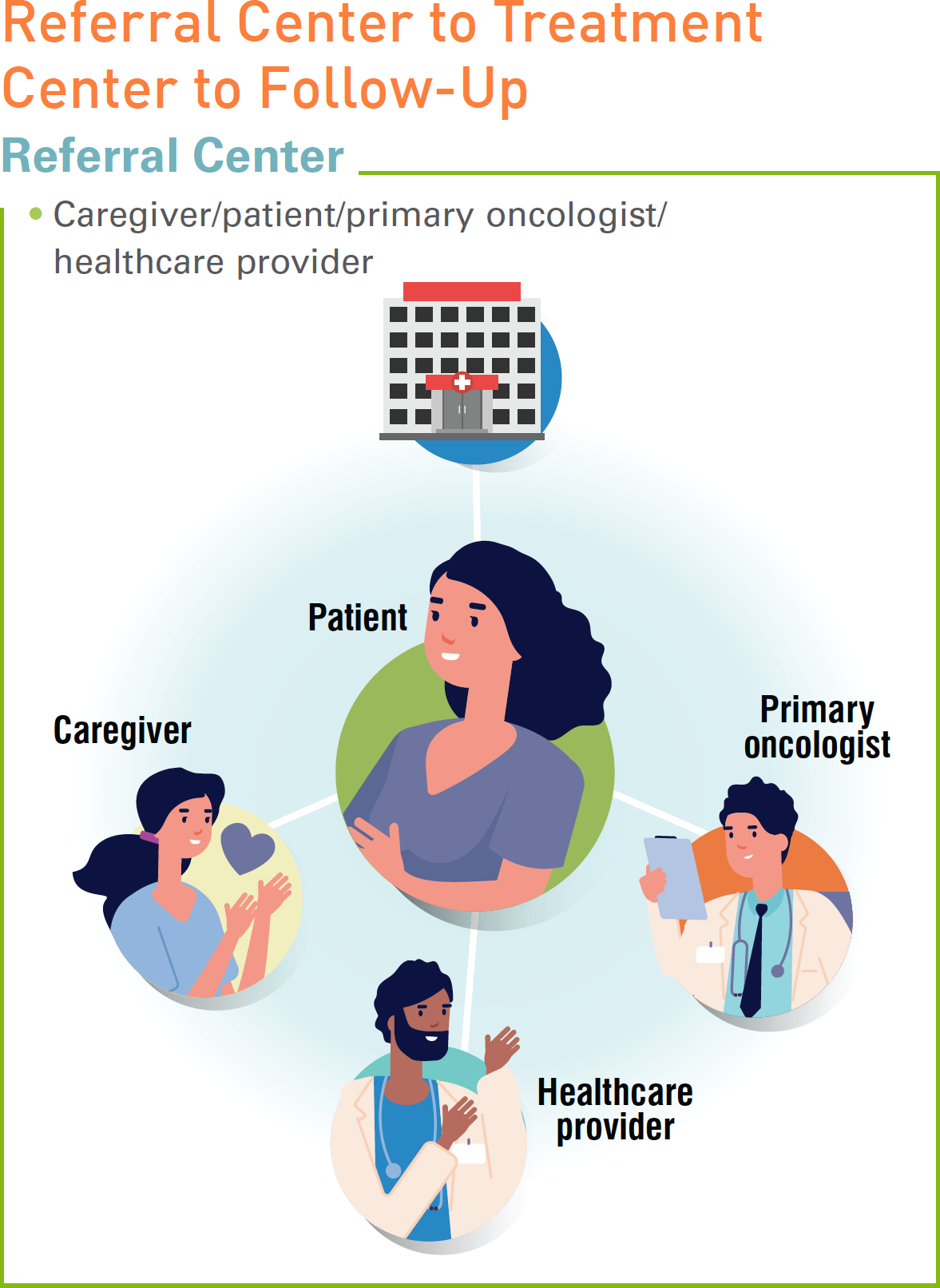
CAR T-cell therapy is a new type of cancer treatment that may be the right option for some patients, particularly those who have failed other therapies. This guide offers best practices for navigators caring for patients undergoing CAR T-cell therapy and their caregivers, from referral through treatment to the follow-up process.
Each phase along a patient’s CAR T-cell journey provides opportunities for navigators to offer support to patients and their caregivers in a variety of ways. This guide is intended to enhance the navigator’s role in supporting the CAR T-cell journey for both patients and their caregivers.
Introduction
Novel cancer therapeutics present challenges for both healthcare professionals and patients, in terms of understanding the clinical applications and the potential impact of a particular treatment on a particular patient.
Navigators play an essential role in helping patients understand new treatments. One of the newest treatments is chimeric antigen receptor (CAR) T-cell therapy.
Evolution of CAR T-Cell Therapy
Immunotherapy has been the focus of investigation for cancer researchers and clinicians since the 1970s. The key premise behind immunotherapy is the potential to use key elements of a patient’s own immune system to fight cancer, through a process called adoptive cell transfer.
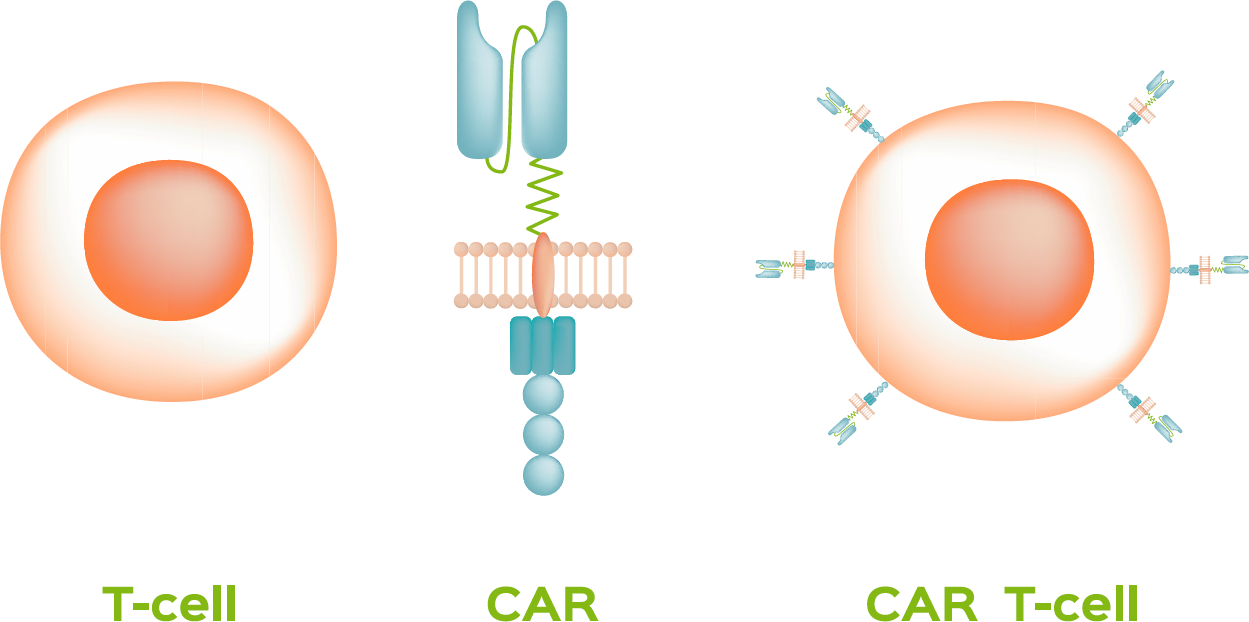
There are several types of adoptive cell transfer. The type that is the farthest along in clinical development is called CAR T-cell therapy. This approach involves collecting blood from a patient and separating its various components using a process known as apheresis. During this process, the laboratory extracts the T-cells from the blood. The T-cells are the white blood cells that fight infection. For a variety of reasons, T-cells do not always “recognize” cancer cells. To correct this problem and recruit the T-cells that are specifically responsible for destroying abnormal cells in the body, scientists alter the T-cells by adding a “chimeric antigen receptor” to their surface—hence, the name CAR T-cell therapy. This receptor is able to latch on to specific antigens on the surface of cancer cells. When it latches on, the T-cell is able to destroy the cancer cells directly or, in some cases, make it easier for the immune system to attack the cancer cells.1
The CAR T-cell treatment process was developed in the late 1980s and early 1990s. Cancer researchers at institutions such as the University of Pennsylvania and Memorial Sloan Kettering Cancer Center (MSKCC) were among the first clinicians to explore how T-cell engineering could be used to fight various solid tumors and blood cancers. In 2002, investigators at MSKCC developed the first effective CAR T-cells for patient use.2 Since that time, cancer researchers have discovered ways to target CAR T-cell therapy against a variety of cancers. With the growing research focused on CAR T-cell therapy, news about its potential is reaching patients with cancer and moving them into the treatment pathway.
Since the initial development of the CAR T-cell strategy, medical scientists have regularly tested the efficacy of the therapy in a wide range of cancers and cancer cell surface antigens. The era of cellular therapy for multiple myeloma has now arrived. Although CAR T-cell therapy currently can be offered only to those patients who have already received several other lines of therapy and have relapsed, the question becomes whether CAR T-cell therapy is incrementally more effective if used much earlier in the course of disease. Additional follow-up and studies are needed to determine the durability of response to CAR T-cell therapy and the best therapeutic approaches for patients whose disease relapses after receiving CAR T-cell treatment.3,4
Overview of Multiple Myeloma
According to the American Cancer Society, multiple myeloma is a cancer of the plasma cells originating in the B-cell line. Plasma cells are a type of white blood cell that is responsible for the production of antibodies that fight infection. Multiple myeloma is a complicated disease in which the cells release too much protein (called immunoglobulin) into the bones and the blood. As multiple myeloma progresses, those plasma cells begin to spill out of the bone marrow and spread throughout the body. As the plasma cells accumulate in the body, they upset the immune system, causing organ damage and the disruption of normal bone formation.5
Approximately 35,000 new cases of multiple myeloma are diagnosed each year. The median age for the development of multiple myeloma is 70 years. The disease is diagnosed more frequently in African Americans than in white Americans and occurs more often in males than in females. The overall 5-year survival rate in patients with multiple myeloma is approximately 54%, which is significantly better than it was in the past. The cause of multiple myeloma remains unknown. Risk factors for the disease include drinking alcohol, obesity, radiation exposure, family history, and exposure to certain chemicals, such as pesticides and herbicides. Although multiple myeloma is an incurable disease, it is highly treatable because of the recent developments of many new therapies. Despite these advances, approximately 12,400 deaths per year are attributed to multiple myeloma.6
Clinically, patients with multiple myeloma may present with no symptoms, with the disease often diagnosed as the result of an incidental finding. At other times, patients may present with back pain, fatigue, and anemia, but multiple myeloma will not usually be the diagnosis that initially comes to mind.7 Multiple myeloma is diagnosed based on blood or urine tests detecting abnormal antibodies, bone marrow biopsies revealing cancerous plasma cells, and radiology findings showing bone lesions. Another common finding is high blood calcium levels. Other symptoms may include recurrent infections, renal dysfunction, and plasmacytomas, which are single lesions of the bone or soft tissue.
Diagnostic testing for multiple myeloma includes blood and urine tests for high calcium levels, low hemoglobin and hematocrit levels, elevated blood urea nitrogen, creatinine levels, and serum and urine protein electrophoresis screening, as well as radiology studies. Blood work may reveal the presence of M-proteins that are produced by myeloma cells. Another abnormal protein produced by myeloma cells is known as beta-2 microglobulin (B2M), which may also be detected in the blood or urine. The presence of B2M can provide clues about the aggressiveness of the disease. A bone marrow biopsy will also be performed, to look for plasma cells in the bone marrow. Specialized tests, such as fluorescence in situ hybridization, can analyze myeloma cells to understand their genetic abnormalities. Tests are also performed to measure the rate at which an individual’s myeloma cells are dividing.8
Radiology studies may be recommended to detect bone issues associated with multiple myeloma. These tests may include x-rays of the skeletal system, magnetic resonance imaging (MRI), computed tomography (CT) scans, or positron emission tomography (PET) scans.
A variety of options are available for the treatment of multiple myeloma. Treatment depends on the stage of the disease, a patient’s performance status, and the goals of treatment. If a patient has monoclonal gammopathy of undetermined significance or smoldering myeloma, observation may be the preferred option, whereas the presence of full-blown multiple myeloma requires treatment.
Treatment options for multiple myeloma may include the following:
- Chemotherapy: Chemotherapeutic agents kill fast-growing cells, including myeloma cells. Chemotherapy drugs can be administered orally or intravenously. Patients receive high doses of chemotherapy drugs prior to undergoing bone marrow transplantation (BMT)
- Corticosteroids: Corticosteroids regulate the immune system, in order to control inflammation in the body. They are also active against myeloma cells. Corticosteroids can be taken orally or administered intravenously
- Stem-cell transplantation: A stem-cell transplantation is a procedure that is performed to replace diseased bone marrow with healthy bone marrow. Prior to a patient undergoing a stem-cell transplantation, blood-forming stem cells are collected from his or her blood. Patients are treated with high doses of chemotherapy to destroy the diseased bone marrow, then stem cells are reinfused into their body, where they travel to the bone marrow and begin the rebuilding process
- CAR T-cell therapy: A type of treatment in which a patient’s T-cells (a type of immune system cell) are modified in the laboratory so they will attack cancer cells. Large numbers of the CAR T-cells are grown in the laboratory and administered to a patient via infusion. CAR T-cell therapy is used to treat certain types of blood cancer and currently is being investigated for the treatment of other forms of cancer as well
- Radiation therapy: This treatment uses beams of energy to damage myeloma cells and halt their growth. Radiation therapy may be used to quickly shrink myeloma cells in a specific area—for example, when a collection of abnormal plasma cells form a tumor (plasmacytoma) that is causing pain or destroying a bone
- Immunotherapy: Immunotherapy uses the body’s immune system to fight cancer; clinical trials with forms of immunotherapy other than CAR T-cell therapy are currently underway
The multiple myeloma treatment paradigm is expanding, with many novel therapies now available to treat relapsed/refractory disease.4
Implications for Navigation Practice
As with other cancer treatments, particularly new therapies, navigators fulfill multiple roles not only in supporting patients, but also in collaborating with a variety of clinical team members who are involved along the treatment continuum.
An article published last year in The Lancet Haematology described the evolution of the roles and responsibilities of nurses and nurse navigators in the delivery CAR T-cell therapy at a single academic oncology center—the Dana-Farber Cancer Institute (DFCI) in Boston. Although the article focuses primarily on the expanding and evolving roles that nurses play in the success of DFCI’s CAR-T therapy programs, it also highlights important information regarding patient navigation programs.9
According to the authors of the article, when the CAR-T therapy program was created in 2016, “The nurse practitioner and nurse navigator roles were developed to focus exclusively on ensuring that safe, high-quality care was provided to these patients through the entire treatment trajectory.”9 The nurse navigator was viewed as a “constant” in the life of the patient—from the initial discussion with the patient about CAR-T therapy, through the patient’s treatment, to assessment of the effects of the treatment and the patient’s response.
What DFCI found upon the introduction of CAR T-cell therapy was that patients and clinical staff required support and education about the treatment. For example, nurses performing apheresis procedures, those delivering outpatient infusions, as well as those working in emergency departments and hospital settings—inpatient oncology nurses, in particular—also required in-depth knowledge on approaches for mitigating the toxicity associated with CAR-T therapy. Understanding all of the steps involved in CAR T-cell therapy, not just the initial step, might also help clinical staff address patient questions that are certain to arise.9
Although the navigators were initially assigned to be patient resources, they also filled important educational resource roles for other nursing team members. Bearing this in mind, patient navigators who are linked to CAR-T therapy programs or who are helping to launch CAR-T therapy programs may find a greater need to enhance their clinical knowledge related to the treatment, so that they can serve as an educational resource for their peers.10
Definition of Patient Navigation
Patient navigation in cancer care is “individualized assistance offered to patients, families, and caregivers to help overcome healthcare system barriers and facilitate timely access to quality health and psychosocial care from prediagnosis through all phases of the cancer experience.”11 Navigation is the process of helping patients overcome health system barriers and providing them with timely access to quality medical and psychosocial care from before their cancer diagnosis through all phases of their cancer experience.12 Navigators help patients and their families manage the challenges of the cancer care continuum, which includes assisting them in processing complex information and making significant decisions along their journey. Patients rely on navigators to act as educators and advocates who can help them feel less overwhelmed by the healthcare system.
There are several roles for navigators in oncology care.13 Nurse navigators are professional registered nurses with oncology-specific clinical knowledge. They act as a liaison between the patient and the healthcare team and provide education and resources to facilitate informed decision-making and timely access to quality health and psychosocial care. Social work navigators are social workers with oncology-specific clinical knowledge. They offer individualized assistance and resources to patients, families, and caregivers to help overcome healthcare system barriers. Patient navigators are patients or nonclinical licensed navigators with a basic understanding of cancer, healthcare systems, and patient access to care. They connect patients to information, resources, and support. The role of navigator in oncology care continues to evolve, with navigators providing specialized information and guidance on the financial aspects of cancer care and treatment with oral oncolytics.
Lessons Learned from a Multiple Myeloma Case Study
The following case study will illustrate the complexity involved in caring for patients with multiple myeloma and the importance of using a multidisciplinary approach to patient management.
Felix is a 62-year-old Spanish-speaking male who presents to his primary care physician (PCP) with back pain. His PCP orders an MRI of the spine, which reveals lytic lesions; blood work is ordered as well. The patient is referred to a cancer center for additional workup and confirmation of a multiple myeloma diagnosis. He is connected to the patient navigator, who begins the initial assessment and triage process. Because of the language barrier, the navigator uses the services of an interpreter for intake. The navigator learns that Felix lives 3 hours away from the cancer center. He is married and has 5 children, aged 20 to 38 years, as well as 4 grandchildren. He is a Jehovah’s Witness. Felix speaks limited English. He previously worked at a factory but had to stop working because of health issues and is on Medicare. His past medical history includes diabetes and hypertension. He takes metformin 1000 mg daily and a thiazide diuretic. Felix tells the navigator that he had been experiencing back pain for several months but did not go to the doctor. He complains of fatigue and decreased “stamina,” indicating that he has “lost his get up and go” and is unable to play with his grandchildren, who bring him a lot of joy. The navigator coordinates an appointment with a medical oncologist and provides Felix with all the details regarding his appointment. The navigator will meet Felix and his family at their medical oncology visit.
The navigator identifies the following barriers to care in establishing a management plan for Felix:
- Logistical: Felix will need help with transportation and lodging if he receives care at the cancer center. Many options are available to Felix and his family. They decide to stay at a Hosts for Hospitals home close to the cancer center. Hosts for Hospitals provides low-cost lodging and support for patients receiving medical care. Volunteer hosts open their homes to patients in need of complex care that is not available close to their home.
- Financial: The patient has Medicare Parts A and B, but no secondary insurance. The navigator knows that Felix will need to secure secondary insurance prior to undergoing a transplant. Felix does not have a prescription plan to cover the costs of his medication. The navigator refers Felix to the financial advocates at the cancer center, who will help him obtain additional insurance. Felix tells his navigator that he thought he had “good insurance” and would not need additional coverage.
- Treatment: The navigator recognizes that communication/education will be a challenge when helping Felix and his family understand the care plan. A decision is made to use interpreter services at all visits. The navigator researches patient educational materials that are available in Spanish to review with Felix. In this case, it also would be appropriate to refer Felix to the bloodless medicine team for a consultation.
- Social: Felix has 5 children and several grandchildren. He has been married to his wife for 40 years, and he tells the navigator that his family is very supportive. Both he and his wife help with the grandchildren, as all of their children and their spouses work.
- Spiritual/Cultural: Felix and his family are Jehovah’s Witnesses. Understanding the beliefs of the Jehovah’s Witness religion will help the multidisciplinary team provide the best care for Felix, by respecting his wishes and understanding his treatment goals. Felix and his family are devout Jehovah’s Witnesses, and they take the tenets of their faith seriously—even if they complicate medical care decisions and even when receiving cancer treatment.
Jehovah’s Witnesses will refuse the transfusion of most blood products, such as packed red blood cells, platelets, white blood cells, and plasma. They allow the transfusion of stem cells and some fractions of blood, such as albumin and cryoprecipitate. (It is possible that Jehovah’s Witnesses will view CAR T-cell therapy as acceptable, given that the infusion involves the patient’s own cells.)
- Communication/Language Barriers: The navigator must make certain that Felix understands his treatment plan, and that both written and oral information is provided in Spanish.
- Emotional/Physical Health: Felix tells his navigator that he has a lot of support, but he is concerned about the impact his illness and his treatment will have on his family. He also expresses the fact that being away from his faith community is a concern.
Care Coordination
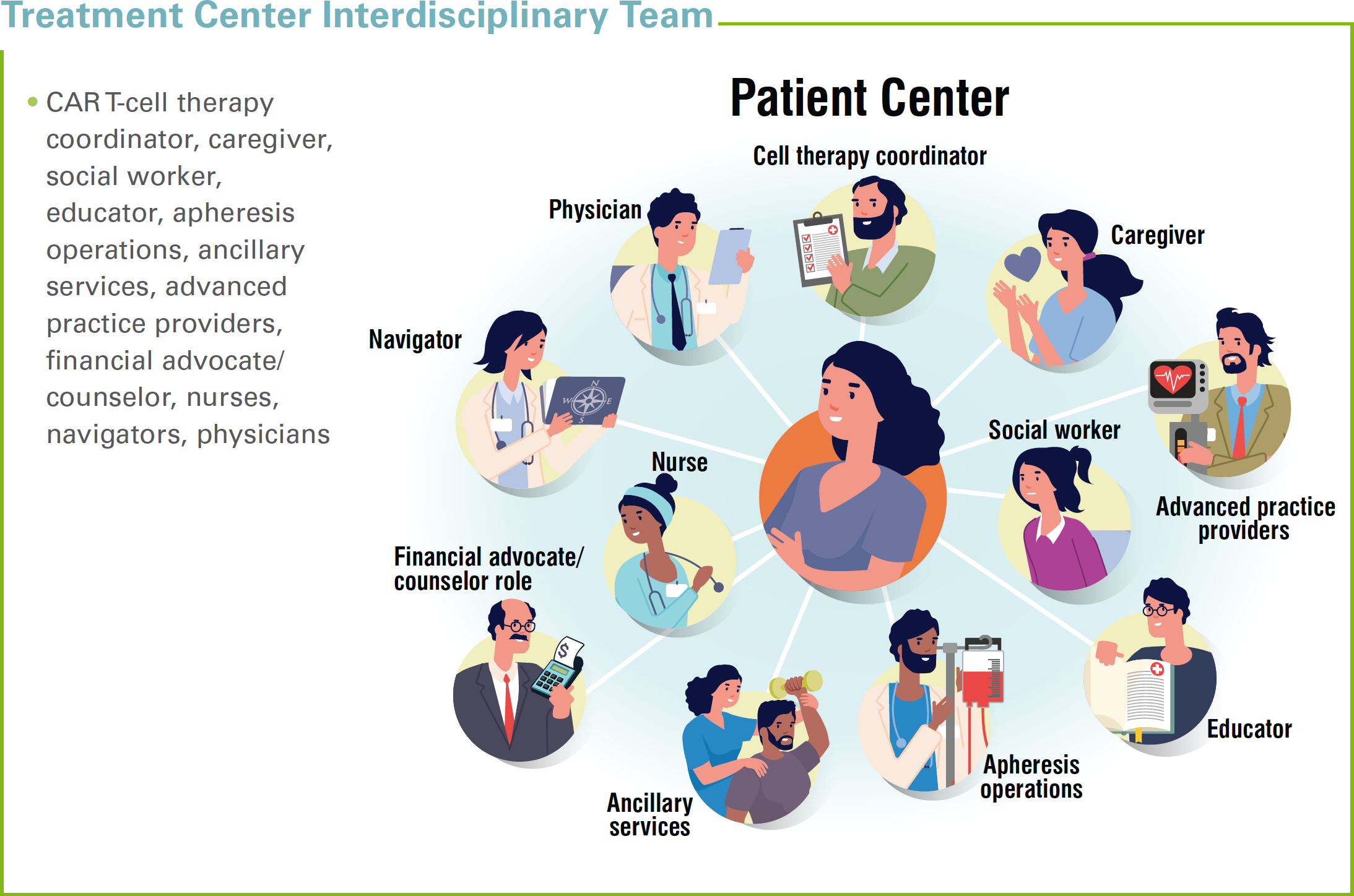
Although it may be tempting to use personnel from the BMT unit to operationalize the CAR T-cell therapy program, differences in patient care and program needs warrant the creation of a CAR T-cell therapy coordinator role. This individual is perched above the clinical team to provide an overview of the traffic flow. The CAR T-cell therapy coordinator can assume the lead in creating program-specific patient education publications, setting documentation standards, and developing discharge planning tools.
The CAR T-cell therapy coordinator can also create the adverse event reporting system, as required by the US Food and Drug Administration (FDA). Since official billing codes do not exist for CAR T-cell therapy, the coordinator may also play an important role in the insurance approval process, working closely with financial coordinators and hospital administrators. The CAR T-cell therapy coordinator will also assist in care coordination with respect to securing follow-up care and urgent care if a patient develops any serious side effects.
Nurse and patient navigators can be critical allies to coordinators. The navigators can help share the patient education material that is developed, coordinate connections with financial resources, and assist with barriers that will slow the initiation of the CAR T-cell therapy process and/or hinder its progress. Both professionals help to ensure patient compliance with therapy and manage the psychosocial needs of patients and caregivers. The nurse and patient navigators can also support shared decision-making processes that are developed to support patients within the CAR T-cell therapy program.
Important Navigation Roles To Support Patients Undergoing CAR-T Therapy
- Follow patient through continuum of care; be that “constant” presence
- Provide appropriate patient education (based on patient culture, literacy) at the various phases of the treatment trajectory
- Evaluate patient for barriers to care
- Advocate for resources, such as financial, housing, transportation (see Financial Toxicity)
- Offer psychosocial support throughout the CAR T-cell process by providing referrals to behavioral health, integrative services, social work, pastoral care, and local and national advocacy resources
- Develop a patient-friendly “itinerary” to enhance patient understanding/compliance with his or her CAR T-cell journey
- Offer patient tours at the treatment center to decrease anxiety regarding apheresis, infusion, and inpatient/outpatient care
- Use distress screening at pivotal points along the CAR-T care continuum
- Integrate navigators with the CAR-T treatment team to enhance general oncology support, education, and resources
- Level-set patient expectations—for example, what to anticipate regarding toxicities—and create individual strategies to help the patient and his or her caregiver occupy time to decrease anxiety
- Engage and educate caregivers in understanding their roles and responsibilities during the patient’s hospital stay, including securing housing and transportation for outpatient care
- Provide patients receiving CAR T-cell therapy and their caregivers with discharge instructions/classes regarding potential management of adverse events and psychological support following discharge
Financial Toxicity
Financial toxicity is a term that is used to describe the toxic impact that cancer, the treatment of cancer, and the recovery from cancer can have on individual and family finances, due primarily to interruptions in work for the patient or other income earners in the family, the direct cost of treatment, and many indirect costs. To help patients who are undergoing CAR T-cell therapy address financial toxicity, referral to the financial assistance team within the healthcare organization, as well as to other external financial assistance agencies that may exist in the community or nationally, is recommended.14 At the initiation of the CAR T-cell process, the financial counselors, advocates, and/or navigators can assist by checking a patient’s medication coverage, home healthcare services, and so forth. In addition, understanding a patient’s goals for care, treatment, and quality of life are important. Financial obstacles should be addressed early on, so that the patient’s care will not be interrupted by the lack of certain financial resources.
CONQUER: the patient voice 5th Annual Patient Guide to Cancer Support Services 2021 (https://conquer-magazine.com/pssguide2021) provides detailed information on financial and other support services offered by pharmaceutical companies, foundations and nonprofit organizations, and government programs. You can also find information on programs for patients and caregivers organized by cancer type, including multiple myeloma.
Patient Issues for Navigators to Consider
Patients who agree to undergo CAR T-cell therapy have a lot to consider. One of the first issues to confront patients is whether they can undergo therapy at the cancer center close to their home or if they will they need to be referred to another center that offers CAR T-cell therapy. Approximately 100 medical centers across the United States offer FDA-approved CAR T-cell therapy, but not every state has a medical center that offers the treatment.15 If a patient meets the criteria for CAR T-cell therapy and must travel far from home for treatment, he or she will require significant caregiver support, which can be costly. The costs most often associated with CAR T-cell therapy include the costs of treatment, such as imaging studies, biopsies, apheresis, CAR T-cell production, and hospital stays. The indirect costs of treatment would include housing and related travel expenses for patients who cannot remain in their home communities for treatment. Because of the lengthy treatment regimen required for CAR T-cell therapy, these indirect costs can be considerable. All of these factors must be considered when navigators are providing information support and resources to patients receiving CAR T-cell therapy.
Referral to CAR T-Cell Therapy
When sharing facts about the CAR-T program, information on FDA-approved CAR-T therapy programs or a clinical trial should be prioritized. Bear in mind that eligibility criteria will differ between commercial CAR-T therapy programs and clinical trials.
First Appointment
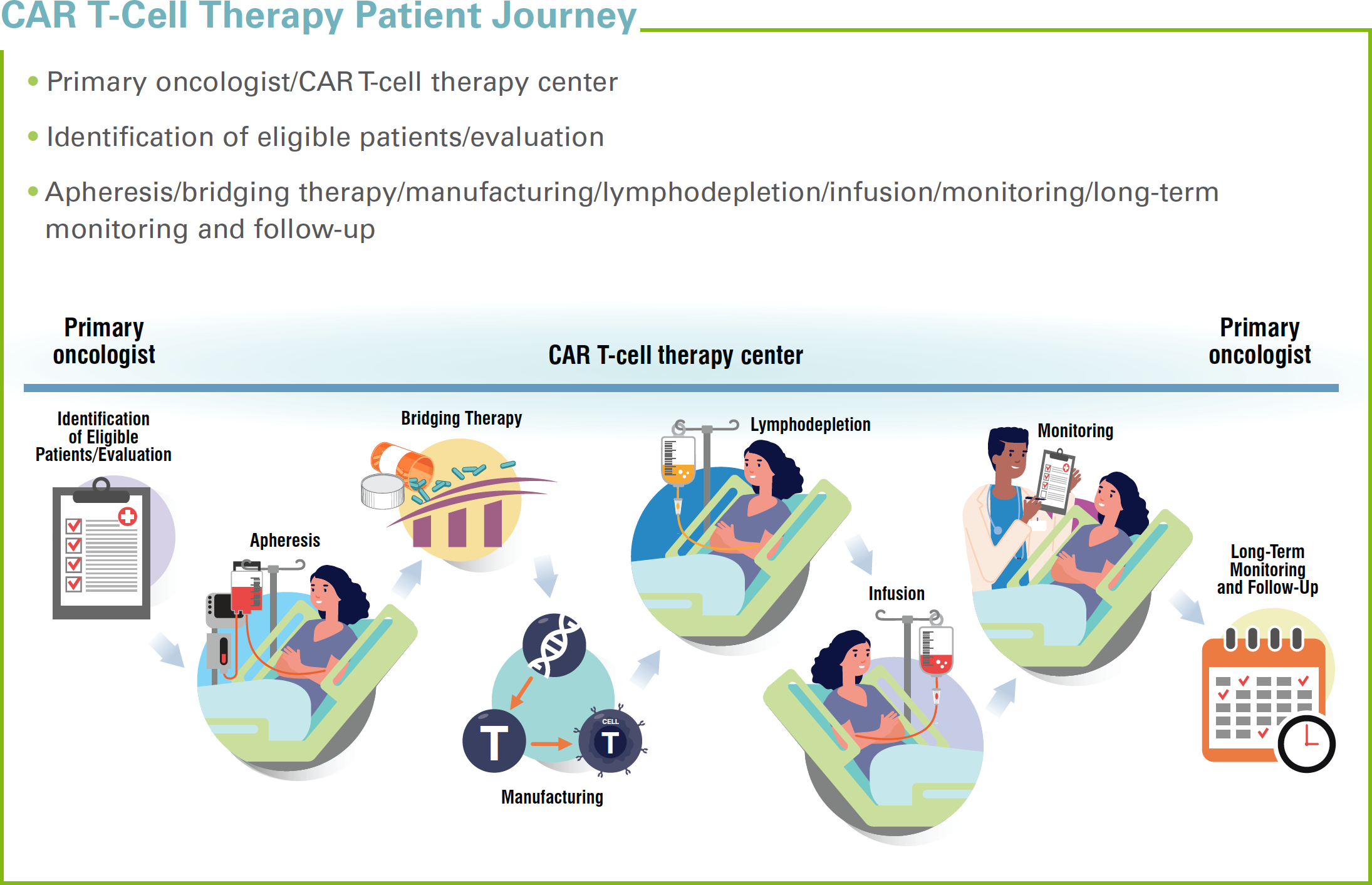
The first appointment for CAR T-cell therapy is critical. During this visit, the patient and the medical team leader for the CAR T-cell program will meet and evaluate whether this form of therapy is truly appropriate for a particular individual. The treatment team will perform screening tests to make certain that the patient is eligible and healthy enough to undergo the treatment.
Screening Tests
Patients need to understand that additional screening tests may be necessary and that multiple appointments may be required after their first visit. Helping patients recognize the need to accommodate a lengthier screening period is probably necessary, so that they and their family members will have realistic expectations and a lower likelihood of experiencing frustration.
Treatments to Qualify for CAR T-Cell Therapy (If Needed)
In certain cases, patients are not physically prepared to undergo CAR T-cell therapy and must receive other treatments to prepare their body, or they must improve their overall health to tolerate the therapy. Although CAR T-cell therapy is perceived as “natural,” it can have side effects, so the healthier a patient is at the start, the better.
When a patient’s cancer cannot be controlled but he or she is still a candidate for CAR T-cell therapy, physicians often recommend the use of bridging therapies, which may cause months-long waits before the actual CAR T-cell infusion therapy process can begin. Even if a patient is healthy right now, he or she may eventually require bridging treatment to stay healthy long enough to proceed to the “infusion” step of CAR-T therapy. Helping patients understand the timing process will help them endure the waiting period that sometimes is involved with CAR T-cell therapy, because of the time it takes to culture sufficient CAR T-cells in the laboratory.
Bridging Therapy (If Needed)
Bridging therapy is cancer treatment that is administered at any step prior to CAR T-cell infusion, to keep the patient healthy enough to get to infusion. Patients may need to receive bridging therapy multiple times throughout their CAR T-cell journey. Each round of bridging therapy can last ≥1 months, but in many cases, it may only span 1 to 2 weeks.
Conditioning Chemotherapy
Prior to CAR T-cell infusion, many patients will undergo conditioning chemotherapy (also known as lymphodepleting chemotherapy), so that their body can accept the new CAR T-cells. This can be confusing for patients, as they may have assumed they were avoiding intense cancer treatments with CAR T-cell therapy. Conditioning chemotherapy most often involves several days of chemotherapy, followed by 2 days of rest. The exact time frame may vary, however, if the patient is in a clinical trial.
Collecting T-Cells for Apheresis
During the apheresis stage of CAR T-cell therapy, the patient will be connected to a machine that separates his or her T-cells from the blood cells. After apheresis, the patient’s T-cells will be sent to the laboratory, where the “chimeric antigen receptor” will be added to the cell surface of the T-cells. The apheresis procedure will take a few hours. Once the cell receptors are added to the T-cells, they will be equipped to find and latch on to the corresponding antigens on the surface of the cancer cell. Once attached, the T-cells can destroy the individual cancer cells. If enough cancer cells are destroyed, the patient may enter remission or be completely cured; alternatively, the therapy may merely serve to add a few more months to the patient’s prognosis.
Waiting Is the Most Difficult Part
Navigators and clinical team members must help patients prioritize patience. Once the T-cells have been harvested, it currently takes several weeks to culture the cells and prepare them for infusion. Patient anxiety during this waiting period can be heightened, so communication is important.
CAR T-Cell Infusion
Once the CAR T-cells are ready, they will be infused into the patient’s bloodstream. The CAR T-cells will begin to find and attack the cancer cells. The infusion can be administered in a hospital or in an outpatient clinic. The infusion appointment may take 20 to 30 minutes, based on the healthcare team and the facility.
Weeks 1 to 8 Following Infusion
Patients who receive CAR T-cell therapy at an inpatient facility may need to stay in the hospital for up to 1 week following the infusion. Patients who receive CAR T-cell therapy on an outpatient basis can typically leave the hospital following the infusion. In either case, patients must be monitored for side effects and should contact their care team immediately if any complications arise. Patients undergoing CAR T-cell therapy are at greatest risk for some type of reaction within the first month following infusion. The patient will need to return to the hospital or clinic if serious side effects develop. For this reason, it is important to help patients understand that they need to make housing arrangements near the medical facility during and after receiving CAR T-cell therapy.
Weeks 9 to 52 Following Infusion
After their treatment has been completed, patients will need to follow up with the CAR T-cell treatment team, their primary oncologist, or other specialists. The follow-up may involve PET and CT scans, to determine whether the therapy worked. If the therapy does appear to be successful, most often, the patient will have monthly laboratory tests or appointments for the first year after infusion. If cleared, the patient may need annual appointments for up to 15 years following CAR T-cell infusion, to check for recurrence, secondary tumors, or late-occurring side effects.
Best Practices for Navigators and CAR T-Cell Therapy Coordinators
Best Practices Prior to and 30 Days Postinfusion
- Provide patient educational resources that are appropriate for language, culture, and level of understanding
- Leverage existing navigation infrastructure/intake and care coordination infrastructure to create efficiencies, rather than creating a host of unique CAR T-cell therapy processes and intake channels
- Include caregivers in all patient educational sessions
- Coordinate handoffs across clinical settings
- Create a CAR T-cell therapy workgroup that includes clinical and administrative personnel
- Because memory loss is a possible late side effect associated with CAR T-cell therapy,14 create a wallet card for the patient to keep with him or her at all times, which will alert any medical providers that the individual has received CAR T-cell therapy (include all key details). This reference card is beneficial to healthcare professionals16
- Ensure that staff policies and processes are unified if the CAR T-cell therapy unit is housed within a larger cancer care unit, such as BMT
- Frequently update external resource lists (eg, housing, transportation options), as the information may change frequently in a dynamic economic region
- Coordinate with the patient’s PCP and other clinical personnel (eg, dentists, psychologists), to ensure that they are aware of the CAR T-cell therapy regimen and its possible side effects
- Educate patients on when and how to contact medical personnel if posttreatment side effects emerge
- Educate the patient’s referring medical team about CAR T-cell treatment side effects and late effects that could emerge even months or years later, so they will not assume the side effects indicate new clinical problems and are prepared for the medical management of side effects
- Leverage existing data and quality management teams, because they are likely well-suited to maintain the data reporting and regulatory monitoring of the CAR T-cell therapy program
- Identify on-site care providers who are dedicated to respond to acute issues associated with the infusion
- Build connections with financial support resources to help patients address cost-of-treatment issues and the need for longer-term housing
Patient Evaluation/Eligibility/Enrollment In CAR T-Cell Therapy Program
- Patients must have a disease that is indicated or being studied in a clinical trial
- Dedicated caregiver
- Ability to stay in close proximity to the treating institution for at least 1 month
- Financial support for available CAR T-cell treatments
- Patient selection may differ by available commercial product
Patient Considerations
- Performance status
- T-cell count
- Organ function
- Accessibility and support
Monitoring and Follow-Up
Monitoring Post 30 Days
Navigators may play a role in supporting the patient in some of these areas:
- Coordinate transition back to primary oncologist
- Response to treatment may be evaluated every 3 months for 2 years
- Monitor patient for persistent neurotoxic symptoms, risk for infection, and prolonged cytopenias
- Antiviral and antibiotic prophylaxis may be recommended
- Management of Pneumocystis jirovecii pneumonia prophylaxis
- Revaccination following CAR T-cell treatment
- Follow individual in the Patient Registry for 15 years
- Lifelong monitoring for secondary malignancies
There are unique considerations for CAR T-cell therapy, including physical, psychosocial, and financial effects. Survivorship data are limited; however, additional research can identify needs that are unique to survivors and assess evidence-based interventions. Many patients who receive CAR T-cell therapy travel long distances for referrals to programs outside their primary setting, requiring collaboration between the referring and treating clinicians to coordinate safe, effective care.17 As indications for CAR T-cell therapy expand, patient management in diverse settings requires an approach and partnership between the referring and treating clinicians that is collaborative and evidence-based. A case report describes the need for open communication among cancer psychiatrists, oncologists, and patients; how communication can impact functional recovery expectations; and the importance of setting goals for recovery among patients receiving CAR T-cell therapy.18 A model of comprehensive care has been proposed in pediatric patients undergoing CAR T-cell therapy, which includes the domains of mutual understanding of goals of care, communication with the referring team, assessment of past/present psychosocial needs, and knowledge/expectations about treatment.19 Navigators can glean lessons from this model to incorporate into adult CAR-T patient care.
Overcoming Barriers to Care
The scope of practice for most navigators includes helping patients address barriers to care; this is true with every type of cancer therapy. For patients undergoing CAR T-cell therapy, the barriers can be numerous. The CAR T-cell clinical regimen requires patients to remain in close proximity to the hospital/clinic where the therapy is being administered, because of the frequency of treatment, monitoring requirements, and importance of rapid intervention should complications arise. In most cases, patients will need to reside near the hospital for at least 1 month, which typically means finding a place to stay, securing reliable transportation for appointments or emergencies, creating a strong but perhaps temporary emotional/physical support network in the community, and figuring out how to “do life” while residing in a new area. The need to live near the hospital could create serious financial obstacles for patients who are already burdened with significant costs for cancer treatment. Compounding this significant stress with potential transportation issues, disruptions in lifestyle, and normal social engagement can create emotional and physical stress that becomes challenging for patients with cancer to manage.
The intensity of CAR T-cell therapy is likely to create barriers to care for nearly every patient, no matter his or her access to financial or social resources. For this reason, it is critical for navigators in CAR T-cell programs to regularly perform barrier assessments. A patient who is experiencing minimal barriers to care at the initiation of treatment might experience an increase in barriers as the treatment progresses.
With this direction in mind, CAR-T navigators should incorporate a standardized barriers to care assessment tool and encourage patients to take the self-assessment, or, even better, they should interview patients using the tool. Although patients may not take the time to perform self-assessments, oftentimes, they may disclose their real struggles when face to face with a navigator.
Overall, the value of advocating on behalf of the patients to overcome barriers related to finances, housing, social support, patient education, and transportation emphasizes the importance of the role played by the navigator who is caring for the patient undergoing CAR T-cell therapy. Navigators must ask and be aware of the common challenges faced by most patients receiving this treatment, and they need to be prepared with resources.
Educate Patients To Seek Emergency Care For The Following Symptoms:
- Fast/irregular heartbeat
- Difficulty breathing
- Fever/chills
- Confusion
- Difficulty speaking
- Difficulty understanding
- language
- Stupor
- Severe fatigue
- Dizziness/low blood pressure
- Gastrointestinal distress
- Severe muscle/joint pain
Caregiver Issues
Just like the patients themselves who are undergoing CAR T-cell therapy, caregivers may find themselves struggling during the active treatment period, as they may be left to focus on the financial challenges involved in finding living quarters, funds for daily living, and building a local network to support their own emotional and physical well-being. Although navigators rightly focus on patients, supporting the caregivers is also a key part of the equation. Caregivers must have access to the information that will help them find housing, easy access to living essentials, and connections in the community that make the cancer treatment period easier. Recommending a caregiver support group/network, as well as providing additional support through financial advocates and/or navigators, will be beneficial for the oncology caregiver. Navigators can contribute to the success of cellular therapy programs by setting appropriate expectations with caregivers regarding such issues as timing of therapy, treatment outcomes, and adverse events.
Clinical Team Issues
Patients undergoing CAR T-cell therapy will encounter many different clinical teams, and care coordination will be an important component in the success of the therapy. As the navigator observes struggles or addresses patient questions in one particular care setting, it will be important for the navigator to communicate with the next care team to appropriately address anxieties or questions that could not be addressed earlier. Acting as a communications bridge for all the respective care teams can help keep patient anxieties or concerns manageable for all the various caregivers who come into contact with the patient.
Program Success Factors
The following success factors have been highlighted in mature navigation programs and should be applicable for navigators involved in CAR T-cell therapy programs20:
- Become as familiar as possible with the clinical pathways, treatment side effects, and other clinical aspects of CAR T-cell therapy
- Identify educational needs for patients and clinical staff, and promote navigation as a resource
- Establish reporting processes to share information on the CAR-T navigation efforts. Consistently using a set of reports to document navigation efforts and disseminating these reports to key stakeholders will build confidence in navigation and elevate the perceived value of navigation across the organization
To support clinical team engagement with the cellular therapy program, nurse and patient navigators can share data on how the program is performing with respect to patient recovery, remission, and cure rates. In many cases, clinical personnel work in silos. This is particularly true when patient care is being delivered in both inpatient and outpatient settings. Clinical personnel involved in cellular therapy may see patients who return because of adverse events, but they may not always hear about the many success stories that are happening as a result of the program. Sharing positive aspects of the program’s impact can boost morale and maintain employee participation, since employee burnout is a common occurrence in many cancer programs.
References
- Carpenter RO, Evbuomwan MO, Pittaluga S, et al. B-cell maturation antigen is a promising target for adoptive T-cell therapy of multiple myeloma. Clin Cancer Res. 2013;19:2048-2060.
- Maher J, Brentjens RJ, Gunset G, et al. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRζ/CD28 receptor. Nat Biotechnol. 2002;20:70-75.
- American Cancer Society. CAR T-cell therapy for multiple myeloma. Revised March 31, 2021. www.cancer.org/cancer/multiple-myeloma/treating/car-t-cell-therapy.html. Accessed September 22, 2021.
- Nebraska Medicine. Holstein SA. CAR T-cell therapy for patients with relapsed/refractory multiple myeloma: ready for prime time. May 11, 2021. www.nebraskamed.com/cancer/treatments/car-t-therapy/car-t-cell-therapy-for-patients-with-relapsed-refractory-multiple-myeloma-ready-for-prime-time. Accessed September 22, 2021.
- American Cancer Society. What is multiple myeloma? Updated February 28, 2108. www.cancer.org/cancer/multiple-myeloma/about/what-is-multiple-myeloma.html. Accessed September 29, 2021.
- Cancer.net. Multiple myeloma. Updated August 2021. www.cancer.net/cancer-types/multiple-myeloma/view-all. Accessed September 29, 2021.
- Leukemia & Lymphoma Society. Signs and symptoms. www.lls.org/myeloma/signs-and-symptoms. Accessed September 30, 2021.
- Leukemia & Lymphoma Society. Diagnosis. www.lls.org/myeloma/diagnosis. Accessed September 30, 2021.
- McDermott K, Spendley L. Nursing impact on the CAR-T cell landscape. Lancet Haematol. 2020;7:e192.
- Oncology Nurse Advisor. Moench S. Roles of nurse practitioner and nurse navigator in a CAR-T therapy program at a large academic oncology center. March 18, 2020. www.oncologynurseadvisor.com/home/cancer-types/hematologic-cancers/roles-of-np-and-nn-in-car-t-therapy-program-at-large-academic-oncology-center. Accessed September 22, 2021.
- National Navigation Roundtable. About patient navigation. https://navigationroundtable.org/about-patient-navigation. Accessed October 7, 2021.
- CoC accreditation requires patient navigation. J Oncol Navig Surviv. 2016;7(8). www.jons-online.com/issues/2016/september-2016-vol-7-no-8?view=article&artid=1478:coc-accreditation-requires-patient-navigation-process. Accessed September 22, 2021.
- Shockney LD, ed. Team-Based Oncology Care: The Pivotal Role of Oncology Navigation. Cham, Switzerland: Springer International Publishing AG, part of Springer Nature; 2018.
- Buitrago J, Adkins S, Hawkins M, et al. Adult survivorship: considerations following CAR T-cell therapy. Clin J Oncol Nurs. 2019;23:42-48.
- BMT Infonet. Medical centers offering CAR T-cell therapy. www.bmtinfonet.org/medical-centers-offering-car-t-cell-therapy. Accessed September 22, 2021.
- Taylor L, Rodriguez ES, Reese A, Anderson K. Building a program: implications for infrastructure, nursing education, and training for CAR T-cell therapy. Clin J Oncol Nurs. 2019;23:20-26.
- HemOnc Today. Jackson J. Access to CAR T-cell therapy faces regional challenges. May 4, 2021. www.healio.com/news/hematology-oncology/20210504/access-to-car-tcell-therapy-faces-regional-challenges. Accessed September 22, 2021.
- Sedhom R, Gupta A, Von Roenn J, Smith TJ. The case for focused palliative care education in oncology training. J Clin Oncol. 2020;38:2366-2368.
- Steineck A, Wiener L, Mack JW, et al. Psychosocial care for children receiving chimeric antigen receptor (CAR) T-cell therapy. Pediatr Blood Cancer. 2020;67:e28249.
- Rowe M, Gentry E. Supporting the value proposition of patient navigation. J Oncol Navig Surviv. 2019;10. www.jons-online.com/issues/2019/february-2019-vol-10-no-2/2245-supporting-the-value-proposition-of-patient-navigation. Accessed September 22, 2021.